Intrathecal baclofen (ITB) is an invasive treatment for spasticity and spasms. A small pump is fitted internally, which can deliver tiny doses of baclofen directly into your spinal fluid. It is useful for people who have spasticity that impacts on their daily lives which is not controlled at tolerable levels of oral medication (pills).
A baclofen pump used to be thought of as a last resort once other treatments stopped being effective. More recently both health professionals and people with MS are realising it can help to keep people mobile and well without the side effects often experienced when medicines are taken as pills.
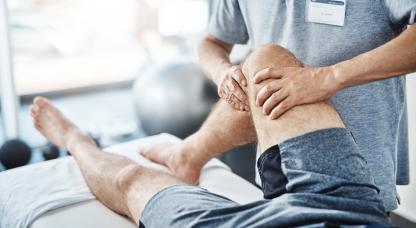
Spasticity means there is an increase in muscle tone. In other words, when the muscle is moved, there is more resistance to this movement than there normally would be. This stiffness can interfere with your fine control causing your movement to be jerky or less smooth.
Spasms are also commonly associated with spasticity. This is when a muscle suddenly contracts, which can cause your arm or leg to suddenly jerk out or flex up towards your body. These spasms can be painful or even dangerous if they occur during transferring or walking.
Spasticity and spasms are a common and troublesome feature of living with MS. In a survey, 84% of 18,727 patients with MS reported at least some symptoms of spasticity, and 30% reported moderate to severe symptoms. If left untreated, severe spasticity can lead to permanent muscle contractures or joint damage.
Spasticity is generally manged through physical exercises. It is important to keep your muscles, ligaments and joints as flexible as possible. A physiotherapist can advise you on how to move and position your body and teach you specific stretches and strengthening exercises.
Urinary problems, constipation, skin breakdown, infection, inappropriate seating, ill fitting clothes or splints can all make your spasticity or spasms worse. It can be very helpful to learn more about your own personal trigger factors. Avoiding these spasticity triggers may prevent the need to take medication.
Sometimes however, exercise and managing trigger factors are not enough and drug treatments also need to be used. Your health professionals will consider your specific experience and any other issues when deciding which drug to try first. For example general stiffness could be treated with baclofen, spasticity with associated neuropathic pain may respond well to gabapentin or pregabalin, and nocturnal spasms disturbing sleep often respond very well to clonazepam.
Whichever drug is used first should be started at a low dose and slowly increased until the goal of treatment is reached. This may be improvement in walking or transfers, less pain or perhaps a good night's sleep. Unfortunately all of the drugs can cause side effects, the commonest of which are drowsiness and light headedness. If side effects are experienced before the goal is reached it may mean a second drug needs to be added in.
What if the tablets don't help?
If it is not possible to control your spasticity or spasms with these drugs without intolerable side effects, Sativex (cannabis extract spray) can be helpful. About half of people with MS and moderate to severe spasticity will respond to Sativex; whether someone is a responder can be identified after a four week trial of the drug. The dose is then controlled by varying the number of sprays taken each day.
Sativex is usually prescribed by an MS or spasticity specialist who will monitor the effects over the trial period and then continue prescribing for those people who have responded. Sativex is approved by NICE, but access remains variable around the UK.
If spasticity and spasms continue to be a problem for you then your health team may consider trying intrathecal baclofen. Intrathecal refers to the space surrounding the spinal cord; this space is filled with cerebrospinal fluid (CSF). Delivering the baclofen into the CSF allows it to act directly on the nerve receptors of the spinal cord. This means tiny doses are used (approximately 100th of the oral dose), thus avoiding the commonly seen side effects of oral baclofen such as drowsiness.
Before fitting a baclofen pump, you'll be invited to have a trial in hospital. Baclofen will be injected into your spinal fluid via a lumbar puncture so you and your team can see how well it works in controlling your spasticity. If you and your team agree that having a pump will help you meet your goals around mobility and comfort, then you can proceed to have one fitted.
The baclofen is delivered by an implanted pump, usually placed in the lower abdomen; this is connected to the intrathecal space via a catheter (very thin tube) which runs under the skin round to the back and into the spine. The baclofen pump is fitted during an operation, usually under general anaesthetic, following a careful assessment of your needs by a multidisciplinary team.
Once fitted, the dose can be adjusted by a programmer held close to the pump outside your body. It can be adjusted to fit with your needs through the day and night to find a routine that gets the best results for you. The pump needs to be refilled via a needle into the pump reservoir at least every 6 months, and may need to be replaced after around six and a half years.
Anyone with troublesome spasticity or spasms that are not helped by oral medication, or who is experiencing side effects, should consider ITB.
In the past, people with MS who were walking were often told "you're not bad enough for that" or "it's not possible to walk with ITB, it will take you off your feet". Although both of these may be true, it is important to discuss this and have a detailed assessment with a neurophysiotherapist and neurologist or rehabilitation specialist. This team can look at whether your spasticity is being best managed and whether it is helping or hindering your mobility.
Often with people who are walking it is impossible to say for sure whether ITB could help until the trial is performed. Around the UK there are many neurological or rehabilitation centres who offer a spasticity service. The first step is to ask to be referred to one of these services.
I’ve had my pump for just over 5 years now. Before it was fitted my leg was very stiff. It felt very heavy, making walking quite hard. I was taking pregabalin and paracetamol three times a day to try and control it. My MS team decided a pump would be useful as I was still working then and we hoped it would make walking easier and keep me mobile.
Before the pump was fitted, I had to have a lumbar puncture with some fluid injected in my spine to see if my legs could tolerate it. That wasn’t nice at all, but my consultant was very good.
I was going to the hospital quite a lot when I first had the pump as we gradually increased the dose to make sure I still had control of my legs. I now go every seven months for a refill.
I still get spasticity in my legs now and again, but it is better than it was. I would definitely recommend the baclofen pump to others. I think sometimes people leave it quite late when they could get the benefit earlier in life.