23 February 2026
How MS is diagnosed: your questions answered
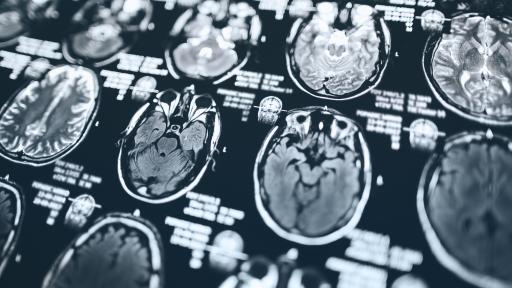
An MS diagnosis can be a long and uncertain process. In this episode, Professor Alasdair Coles explains how neurologists piece things together, from first symptoms to advanced scans, to reach a diagnosis and guide people through what happens next.